About Us
Humanity in our DNA
We Are Focused In Developing Innovative Products
SSPL has for the first time in the world, launched a generic drug where the treatment cost of the brand product was USD 25000 and only available in the US for over 20 years. We have made the generic available in India and in several countries at approximately 1200 USD (5% of the originator). SSPL also holds patents on diseases related to kidney where presently there are no medicines available to suffering patients.
What People Say About Us

Mr. Narayan Jajodia was very supportive and the first imports by us in 1988 were indented by him. Subsequently he encouraged in expanding the product range and supplied key APIs at reasonable terms and arranging meeting with suppliers at various pharma trade fairs internationally.
Dr. Rajendra Agarwal

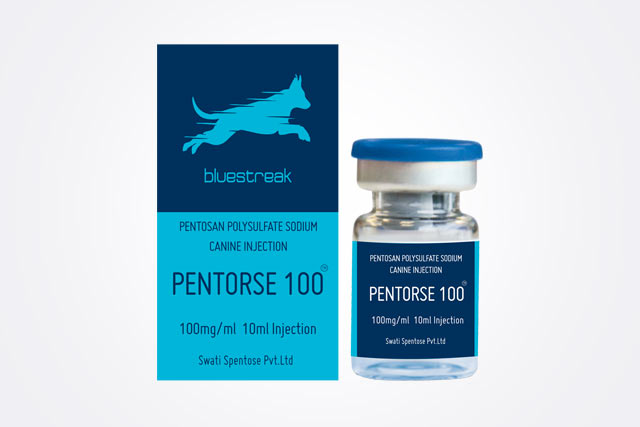
Mr. Vishal deserves to be appreciated for his creative, innovative and industrious attitude and approach. His vision and dream to play globally has come true with launching of the generic molecule Pentosan Polysulphate Sodium globally.
Mr. Binish Chudgar

I am really glad that your company has taken up the commercial production of the drug Pentosan Polysulphate Sodium. This is indeed very commendable achievement of which you can certainly be proud of. All of us in the industry are happy for you and hope that you can expand vour manufacture in the complex area of carbohydrate chemistry
Dr. Y. K. Hamied

Dear Vishal, I extend my congratulations to you and the entire team for the recent successful US FDA inspection. With the foundation that was laid by Late Mr. Narayan Jajodia, our relationship goes back over a few decades. The high degree of commitment which you have shown to grow your father’s business is really commendable.
Mr. Dilip Shanghvi

Why People Trust Us
High Quality Lab
Unmatched Expertise
Precise Result
Qualified Staff

“Ms. Swati Jajodia is the head of Swati Spentose’ s key marketing efforts, responsible for the meteoric rise of the company. ”
– Ms. Swati V. Jajodia, Chief Executive Officer
Our Facilities

SSPL Unit I
USFDA inspected, WHO GMP certified, COFEPRIS Mexico and Korean MFDS approved, AFM available.

SSPL Unit II
WHO-GMP Certified & PMDA Japan Approved

SSPL Unit III
Formulations Site
We Are Certified